Advancing botanical safety: A strategy for selecting, sourcing, and characterizing botanicals for developing toxicological tools
- Publication Date :
- Publication Type : Journal Article
- Author(s) : Waidyanatha et al.
- Journal Name : Food and Chemical Toxicology
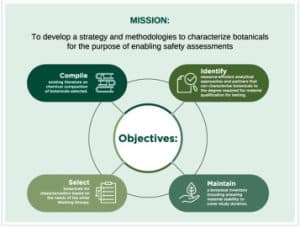
Increases in botanical use, encompassing herbal medicines and dietary supplements, have underlined a critical need for an advancement in safety assessment methodologies. This manuscript highlights: 1) Botanical Safety Consortium's strategy for botanical selection, sourcing, and preparation of extracts to be used in in vitro assays, and 2) the approach utilized to characterize botanical extracts, using green tea and Asian ginseng as examples, to build confidence for use in biological assays.
Advancing botanical safety: A strategy for selecting, sourcing, and characterizing botanicals for developing toxicological tools. Waidyanatha et al., February 2024. Food and Chemical Toxicology. https://doi.org/10.1016/j.fct.2024.114537
Learn more about the Botanical Safety Consortium here.