HESI EnviroTox Database Lauded in European Commission Science Update

The EnviroTox Database, an international project led by the HESI Animal Alternatives in Environmental Risk Assessment Committee, was highlighted in the recent European Commission Science Update, “New Online Tool to Improve the Environmental Risk Assessment of Chemicals”. The EnviroTox Database is a public database of ecotoxicological data which brings together more than 91,000 curated records for more than 4,000 chemicals across 1,500 species. The European Commission called the EnviroTox Database “a valuable resource”, considering it a “credible way of filling data gaps”. Read the full Science Update here.
PATB & COMPARE at 2019 International Symposium on Molecular Allergology

The HESI PATB Committee and COMPARE Allergen Database were well represented at the 2019 International Symposium on Molecular Allergology (ISMA), a focused meeting of the European Academy of Allergy and Clinical Immunology (EAACI) held in Amsterdam, The Netherlands, from 28-30 November 2019. A poster was presented highlighting the latest work of PATB novel research efforts, analyzing the impact of food matrices on protease resistance of strawberry and peach allergens during in-vitro digestibility assays. Additionally, the COMPARE Allergen Database was highlighted as part of two talks by Prof. Lars Poulsen and Dr. Karin Hoffmann-Sommergruber in a focused session on the molecular basis of cross-reactivity.
We gratefully thank PATB Committee & COMPARE expert panel members Prof. Ronald van Ree (Academic Medical Center, University of Amsterdam) and Prof. Lars Poulsen (Copenhagen University Hospital at Gentofte), as well as COMPARE expert panel member Dr. Karin Hoffmann-Sommergruber (Medical University of Vienna, Austria) for their contributions to these programs!
CT-TRACS Publication in Cytotherapy
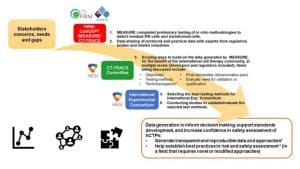
Congratulations to the HESI CT-TRACS Committee for reaching a major milestone with the publication of the Tumorigenicity Working Group’s position paper in Cytotherapy journal last month, “Tumorigenicity Assessment of Cell Therapy Products: The Need for Global Consensus and Points to Consider” (doi: https://doi.org/10.1016/j.jcyt.2019.10.001; open access). This effort brings together stakeholders from five countries, 11 organizations, and many collaborators from various government agencies, cell therapy centers of excellence, cell therapy developers, CROs, and enabling tools providers. The article also describes the working group’s new initiative to generate data evaluating in-vitro test methods through an international multi-site study. As one external expert pointed out, “The international collaboration described in the paper is essential to the translation of the results into widely accepted guidelines in the future, and the initiative taken to develop these data is to be commended.”
Cuba, Cardio-Oncology, and HESI

HESI’s Executive Director, Dr. Syril Pettit, was the inaugural speaker at the 3rd Annual Cuban Conference on Cardio-Oncology held in Havana, Cuba from 12-14 December 2019. HESI co-sponsored the conference. Dr. Pettit addressed a group of nearly 200 cardiologists and oncologists with a focus on HESI’s/her research in cardiac safety and opportunities to enhance long term cancer patient quality of life through improved stakeholder collaboration and research. The event, HESI’s first engagement in Cuba, provided an exciting opportunity to build new partnerships with outstanding Cuban scientists and clinicians dedicated to improved cancer patient outcome.
Engaging Stakeholders in
Environmental Epidemiology

On 6 December 2019, the HESI Environmental Epidemiology Committee held a focus group in Atlanta, GA. This meeting was successful in engaging local and federal government regulators (Georgia Department of Health, US Centers for Disease Control / Agency for Toxic Substances and Disease Registry), academic researchers (Emory University), and industry scientists (American Chemistry Council) in an open dialogue on the use of epidemiology and exposure science in risk assessment and decision making. This was the fourth meeting in a series of highly interactive workshops (two were organized in 2018 in Washington, DC, and Research Triangle Park, NC, as well as one at the Gulf Coast Consortia in Houston, TX, in October 2019) by which we sought to explore incentives and barriers to realizing the full potential of epidemiological data in risk assessment. Starting in early 2020, the Committee will be involved in the development of actionable steps aiming to foster change in the field, with the ultimate goal to leverage and synergize the efforts of epidemiologists, exposure scientists, and risk assessors to improve public health. For more information, please contact Sandrine Deglin (sdeglin@hesiglobal.org).
Emerging Issues 2020
Call for Proposals – Now Open!
![]()
HESI is seeking your suggestions for new scientific projects (human or environmental health) for consideration as an emerging issue in 2020. Proposals will be reviewed in early 2020, and one or more will be selected to form a new scientific collaborative program within HESI. Although this is not a grant, the selected program will receive support from HESI for scientific program design, coordination, and staffing.
Learn more about the HESI Emerging Issues Process on our website or download the proposal form here. The deadline to submit proposals is 31 January 2020.
You can also submit ideas any time of the year via the HESI online Open Forum! Click here to learn more.
Congratulations to former
HESI Board of Trustees Members!

HESI congratulates Dr. Sam Cohen (University of Nebraska Medical Center) and Dr. Jim Klaunig (Indiana University) for being elected as AAAS Medical Sciences Fellows, as well as Dr. Jay Goodman (Michigan State University) for being elected as an AAAS Pharmaceutical Sciences Fellow! Both Dr. Cohen and Dr. Goodman are former Chairs of the HESI Board of Trustees, and Dr. Klaunig is a former HESI Board of Trustees member and current HESI Emerging Issues Committee member. The 2019 group of AAAS Fellows will receive official certificates and rosette pins in a ceremony on 15 February 2020 during the AAAS Annual Meeting in Seattle, WA. View the full list of AAAS Fellows on the AAAS website.
Happy Holidays from HESI!

Happy Holidays and Happy New Year! Thank you to everyone who helped make 2019 successful for HESI. We wish you all a happy holiday season and a healthy and joyful New Year.
Save the Date! Cancer Immunotherapies Training Course

The HESI Immuno-safety Technical Committee (ITC) will hold a training course in Tres Cantos, Spain, on 22–23 April 2020 on the topic of cancer immunotherapies. This 2-day course will provide the foundation biology, describe therapeutic approaches across the various modalities, and discuss other safety-related issues. For more information, please visit the website or contact Stan Parish (sparish@hesiglobal.org).
From the Leadership

Syril D. Pettit, DrPH, MEM
HESI Executive Director